
Chemistry, 02.03.2020 16:56 raynaesquivel
A reaction mixture initially contains 0.86 atm NO and 0.86 atm SO3. Determine the equilibrium pressure of NO2 if Kp for the reaction at this temperature is 0.0118. NO(g) SO3(g) NO2(g) SO2(g)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1
You know the right answer?
A reaction mixture initially contains 0.86 atm NO and 0.86 atm SO3. Determine the equilibrium pressu...
Questions




Physics, 09.10.2019 06:30

Mathematics, 09.10.2019 06:30


Geography, 09.10.2019 06:30

Mathematics, 09.10.2019 06:30



Mathematics, 09.10.2019 06:30


Mathematics, 09.10.2019 06:30

Health, 09.10.2019 06:30


History, 09.10.2019 06:30


Geography, 09.10.2019 06:30


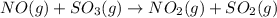
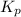 = 0.0118.
= 0.0118.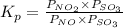
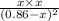
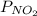 is 0.084 atm.
is 0.084 atm.

