Chemistry, 02.03.2020 16:51 vladisking888
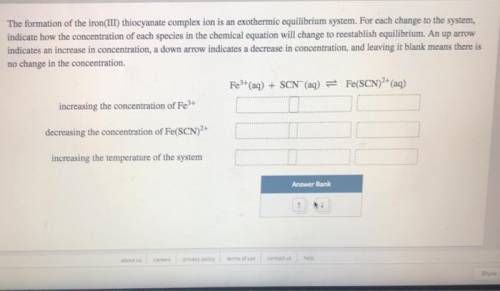
The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change to the system, indicate how the concentration of each species in the chemical equation will change to reestablish equilibrium. An up arrow indicates an increase in concentration, a down arrow indicates a decrease in concentration, and leaving it blank means there is no change in the concentration.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:50
Which of these reactions are redox reactions? check all that apply.cd + hcl → cdcl2 + h2cucl2 + na2s → 2nacl + cuscaco3 → cao + co2 2zns + 3o2 → 2zno + 2so2 ch4 + 2o2 → co2 + 2h2o
Answers: 3

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3


Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
You know the right answer?
The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each...
Questions


History, 29.06.2019 17:30



Physics, 29.06.2019 17:30


Mathematics, 29.06.2019 17:30

Biology, 29.06.2019 17:30





Mathematics, 29.06.2019 17:30


Health, 29.06.2019 17:30

Mathematics, 29.06.2019 17:30

History, 29.06.2019 17:30


Mathematics, 29.06.2019 17:30

Mathematics, 29.06.2019 17:30