
Chemistry, 02.03.2020 18:29 mariahisawsome1
Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite mineral rhodochrosite, involves a two-step process. In the first step, manganese (II) carbonate and oxygen react to form manganese (IV) oxide and carbon dioxide: 2 MnCO3(s) + O2(g) → 2MnO2(s) + 2CO2(g) In the second step, manganese (IV) oxide and aluminum react to form manganese and aluminum oxide: 3 MnO2(s) + 4 Al(s) → 3 Mn(s) + 2 Al2O3(s) Write the net chemical equation for the production of manganese (II) carbonate, oxygen and aluminum. Be sure your equation is balanced.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3
You know the right answer?
Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite min...
Questions





English, 24.10.2020 18:10


Mathematics, 24.10.2020 18:10


Computers and Technology, 24.10.2020 18:10


Mathematics, 24.10.2020 18:10


Mathematics, 24.10.2020 18:10


History, 24.10.2020 18:10

Mathematics, 24.10.2020 18:10


English, 24.10.2020 18:10

Arts, 24.10.2020 18:10

Arts, 24.10.2020 18:10

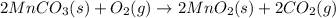
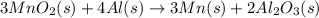
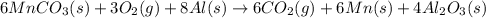
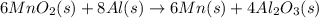
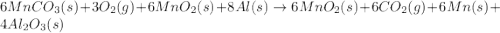
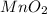 is common on both side, by cancelling it, we get:
is common on both side, by cancelling it, we get:

