
Chemistry, 02.03.2020 22:59 hoopstarw4438
At a certain temperature, 3.67 mol SO 2 and 1.83 mol O 2 are placed in a container. 2 SO 2 ( g ) + O 2 ( g ) − ⇀ ↽ − 2 SO 3 ( g ) At equilibrium, there is 1.92 mol SO 3 present. Determine the number of moles of SO 2 and O 2 that are present when the reaction is at equilibrium.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 21:00
Read "who built the pyramids? ”. leave this link open while you answer the questions throughout the assignment. give at least two reasons why some people claim the pyramids of giza were constructed by aliens.
Answers: 1

Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
You know the right answer?
At a certain temperature, 3.67 mol SO 2 and 1.83 mol O 2 are placed in a container. 2 SO 2 ( g ) + O...
Questions


Mathematics, 20.09.2020 01:01


Mathematics, 20.09.2020 01:01

Physics, 20.09.2020 01:01

Mathematics, 20.09.2020 01:01







History, 20.09.2020 01:01

English, 20.09.2020 01:01


Mathematics, 20.09.2020 01:01

Business, 20.09.2020 01:01

English, 20.09.2020 01:01

Mathematics, 20.09.2020 01:01

 and
and  at equilibrium is, 1.75 mol and 0.87 mol respectively.
at equilibrium is, 1.75 mol and 0.87 mol respectively.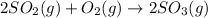
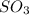 at equilibrium = 1.92
at equilibrium = 1.92


