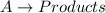Consider a simple reaction in which a reactant AA forms products: A→productsA→products What is the rate law if the reaction is zero order with respect to AA? First order? Second order? For each case, explain how a doubling of the concentration of AA would affect the rate of reaction.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1


Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
Consider a simple reaction in which a reactant AA forms products: A→productsA→products What is the r...
Questions



Mathematics, 21.11.2019 08:31


Mathematics, 21.11.2019 08:31

History, 21.11.2019 08:31


Mathematics, 21.11.2019 08:31



English, 21.11.2019 08:31

History, 21.11.2019 08:31

Social Studies, 21.11.2019 08:31


English, 21.11.2019 08:31


Social Studies, 21.11.2019 08:31

Chemistry, 21.11.2019 08:31

Social Studies, 21.11.2019 08:31

![Rate=k[A]^0](/tpl/images/0531/2473/c3515.png)
![Rate=k[A]^1](/tpl/images/0531/2473/044b8.png)
![Rate=k[A]^2](/tpl/images/0531/2473/e6b6e.png)