
Chemistry, 03.03.2020 01:25 KenzieD7876
H2(g) + F2(g)2HF(g) Using standard thermodynamic data at 298K, calculate the entropy change for the surroundings when 2.20 moles of H2(g) react at standard conditions. S°surroundings = J/K

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
H2(g) + F2(g)2HF(g) Using standard thermodynamic data at 298K, calculate the entropy change for the...
Questions

Mathematics, 11.01.2022 16:30

Mathematics, 11.01.2022 16:30

Social Studies, 11.01.2022 16:30

Mathematics, 11.01.2022 16:30

Mathematics, 11.01.2022 16:40



Geography, 11.01.2022 16:40




English, 11.01.2022 16:50

Mathematics, 11.01.2022 16:50


Mathematics, 11.01.2022 16:50



History, 11.01.2022 16:50


English, 11.01.2022 16:50

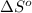 for the surrounding when given amount of hydrogen gas is reacted is -31.02 J/K
for the surrounding when given amount of hydrogen gas is reacted is -31.02 J/K![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0531/3518/52737.png)
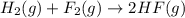
![\Delta S^o_{rxn}=[(2\times \Delta S^o_{(HF(g))})]-[(1\times \Delta S^o_{(H_2(g))})+(1\times \Delta S^o_{(F_2(g))})]](/tpl/images/0531/3518/0a21f.png)

![\Delta S^o_{rxn}=[(2\times (173.78))]-[(1\times (130.68))+(1\times (202.78))]\\\\\Delta S^o_{rxn}=14.1J/K](/tpl/images/0531/3518/7b4e1.png)



