Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1

Chemistry, 23.06.2019 08:00
Technician a says that you should never jump-start a frozen battery. technician b says that a frozen battery can explode, causing injury, when jump-started. who is correct?
Answers: 2

Chemistry, 23.06.2019 11:00
The lab procedure involves several factors, listed below some were variable and some were constant. label each factor below v for variable ot c for constant
Answers: 1
You know the right answer?
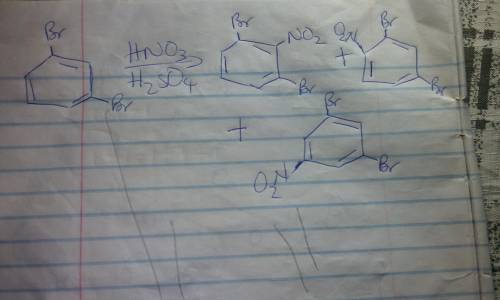
A turn-of-the-century chemist isolated an aromatic compound of molecular formula C6H4Br2. He careful...
Questions


Mathematics, 09.10.2019 11:00

Mathematics, 09.10.2019 11:00

Mathematics, 09.10.2019 11:00




Geography, 09.10.2019 11:00

Mathematics, 09.10.2019 11:00


Chemistry, 09.10.2019 11:00

Social Studies, 09.10.2019 11:00


Mathematics, 09.10.2019 11:00


Mathematics, 09.10.2019 11:00

History, 09.10.2019 11:00

Health, 09.10.2019 11:00