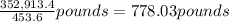Chemistry, 03.03.2020 05:43 freshysans4
A metallurgical firm wishes to dispose of 1200 gallons of waste sulfuric acid whose molarity is 1.05 M. Before disposal, it will be reacted with calcium hydroxide (slaked lime), which costs $0.25 per pound. Write the balanced chemical equation for this process. (Use the lowest possible coefficients. Use the pull-down boxes to specify states such as (aq) or (s). If a box is not needed, leave it blank.) Determine the cost that the firm will incur from this use of slaked lime. Cost

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3
You know the right answer?
A metallurgical firm wishes to dispose of 1200 gallons of waste sulfuric acid whose molarity is 1.05...
Questions

History, 30.06.2019 14:30

Mathematics, 30.06.2019 14:30




Mathematics, 30.06.2019 14:30

Biology, 30.06.2019 14:30



English, 30.06.2019 14:30

Computers and Technology, 30.06.2019 14:40

Mathematics, 30.06.2019 14:40


Social Studies, 30.06.2019 14:40




Mathematics, 30.06.2019 14:40


History, 30.06.2019 14:40

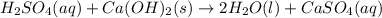
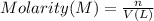
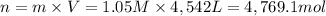
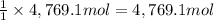 of calcium hydroxide
of calcium hydroxide