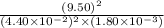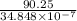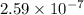Chemistry, 03.03.2020 06:14 alejandra216
Be sure to answer all parts. The equilibrium constant (Kc) for the formation of nitrosyl chloride, an orange-yellow compound, from nitric oxide and molecular chlorine 2NO(g) + Cl2(g) ⇌ 2NOCl(g) is 1 × 107 at a certain temperature. In an experiment, 4.40 × 10−2 mole of NO, 1.80 × 10−3 mole of Cl2, and 9.50 moles of NACl are mixed in a 2.60−L flask. What is Qc for the experiment

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
Be sure to answer all parts. The equilibrium constant (Kc) for the formation of nitrosyl chloride, a...
Questions


Mathematics, 07.10.2019 01:30

History, 07.10.2019 01:30

Chemistry, 07.10.2019 01:30


History, 07.10.2019 01:30

English, 07.10.2019 01:30


History, 07.10.2019 01:30

Biology, 07.10.2019 01:30

History, 07.10.2019 01:30




Geography, 07.10.2019 01:30



Social Studies, 07.10.2019 01:30

History, 07.10.2019 01:30

Mathematics, 07.10.2019 01:30

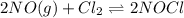
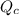 of this reaction is as follows.
of this reaction is as follows.![Q_{c} = \frac{[NOCl]^{2}}{[NO]^{2}[Cl_{2}]}](/tpl/images/0532/0245/90950.png)