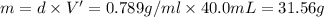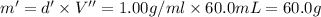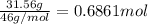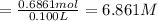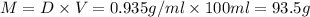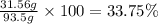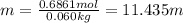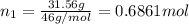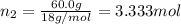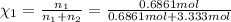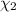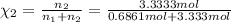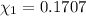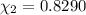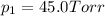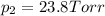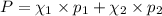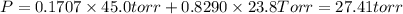Chemistry, 04.03.2020 02:22 bgallman153p71edg
Grey Goose ® vodka has an alcohol content of 40.0 % (v/v). Assuming that vodka is composed of only ethanol and water answer the following questions. Note: The molar masses of water and ethanol are 18.0 g and 46.0 g, respectively. The densities of water, ethanol, and this vodka mixture are 1.00 g/mL, 0.789 g/mL, and 0.935 g/mL, respectively
a. Calculate the molarity of ethanol in this vodka, assuming that water is the solvent.
b. Calculate the percent by mass of ethanol % (m/m) in this vodka.
c. Calculate the molality of ethanol in this vodka assuming that water is the solvent.
d. Calculate the mole fractions of ethanol and water in this vodka.
e. Calculate the vapor pressure, in torr, of this vodka at 25.0 oC if the vapor pressures of pure water and ethanol are 23.8 torr and 45.0 torr, respectively?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2


Chemistry, 23.06.2019 09:00
Need ! assume that the variables x and y are directly related. if k = 8, what is the value for each of the following points? be sure and record your data to be used in the following problem. x y k 0.
Answers: 2

You know the right answer?
Grey Goose ® vodka has an alcohol content of 40.0 % (v/v). Assuming that vodka is composed of only e...
Questions



Business, 30.10.2019 14:31






Social Studies, 30.10.2019 14:31

Geography, 30.10.2019 14:31




Health, 30.10.2019 14:31


History, 30.10.2019 14:31


Mathematics, 30.10.2019 14:31

Mathematics, 30.10.2019 14:31