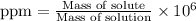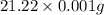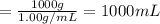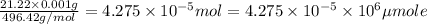Chemistry, 04.03.2020 02:57 flowersthomas1969
Calculate and report the precise concentration of undiluted stock standard solution #1 for AR in micromoles per liter from ppm by mass. Assume that the density of water is 1.00g/ml. This is your most concentrated undiluted standard solution for which you measured the absorbance.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 22.06.2019 23:00
Arectangle has a diagonal 20 inches long that forms angles of 60 and 30 with the sides. find the perimeter of the rectangle. for geometry
Answers: 3

Chemistry, 23.06.2019 02:00
The bone of a dinosaur and the imprint of a leaf are examples of which kind of fossils? a) index b) body c) amber d) trace
Answers: 1

Chemistry, 23.06.2019 02:20
Which of the following will cause an increase in the acceleration of an object? increase force decrease force increase mass decrease mass
Answers: 1
You know the right answer?
Calculate and report the precise concentration of undiluted stock standard solution #1 for AR in mic...
Questions

Mathematics, 31.03.2021 17:50


Mathematics, 31.03.2021 17:50





English, 31.03.2021 17:50

Business, 31.03.2021 17:50

English, 31.03.2021 17:50

Mathematics, 31.03.2021 17:50


Mathematics, 31.03.2021 17:50



Mathematics, 31.03.2021 17:50

Mathematics, 31.03.2021 17:50

Mathematics, 31.03.2021 17:50


Mathematics, 31.03.2021 17:50