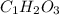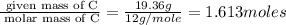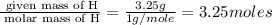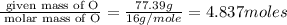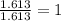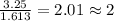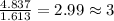Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:10
The rock in a lead ore deposit contains 89 % pbs by mass. how many kilograms of the rock must be processed to obtain 1.5 kg of pb?
Answers: 1

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1
You know the right answer?
What is the empirical formula of a compound composed of 3.25% hydrogen ( H ), 19.36% carbon ( C ), a...
Questions


Mathematics, 21.11.2019 12:31

Mathematics, 21.11.2019 12:31

Computers and Technology, 21.11.2019 12:31

History, 21.11.2019 12:31



Computers and Technology, 21.11.2019 12:31

English, 21.11.2019 12:31

Social Studies, 21.11.2019 12:31


History, 21.11.2019 12:31


Mathematics, 21.11.2019 12:31


Mathematics, 21.11.2019 12:31

SAT, 21.11.2019 12:31

Mathematics, 21.11.2019 12:31