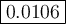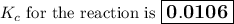Calculate the value of the equilibrium constant, K c , for the reaction Q ( g ) + X ( g ) − ⇀ ↽ − 2 M ( g ) + N ( g ) given that M ( g ) − ⇀ ↽ − Z ( g ) K c 1 = 3.15 6 R ( g ) − ⇀ ↽ − 2 N ( g ) + 4 Z ( g ) K c 2 = 0.509 3 X ( g ) + 3 Q ( g ) − ⇀ ↽ − 9 R ( g ) K c 3 = 12.5

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
What pressure will be exerted by 0.675 moles of a gas at 25*c if it is in a 0.750-l container?
Answers: 1

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

You know the right answer?
Calculate the value of the equilibrium constant, K c , for the reaction Q ( g ) + X ( g ) − ⇀ ↽ − 2...
Questions


Mathematics, 05.12.2021 21:10

Chemistry, 05.12.2021 21:10

Spanish, 05.12.2021 21:10

Mathematics, 05.12.2021 21:10

Mathematics, 05.12.2021 21:10


English, 05.12.2021 21:10

Social Studies, 05.12.2021 21:20


English, 05.12.2021 21:20






History, 05.12.2021 21:20

Mathematics, 05.12.2021 21:20

Social Studies, 05.12.2021 21:20

Chemistry, 05.12.2021 21:20