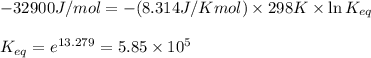Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3

Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2
You know the right answer?
Using any data you can find in the ALEKS Data resource, calculate the equilibrium constant at for th...
Questions


History, 13.12.2020 09:50

Mathematics, 13.12.2020 09:50







English, 13.12.2020 09:50


Mathematics, 13.12.2020 09:50

Mathematics, 13.12.2020 09:50

Mathematics, 13.12.2020 09:50

Mathematics, 13.12.2020 09:50

Geography, 13.12.2020 09:50


Mathematics, 13.12.2020 09:50


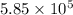
![\Delta G^o_{rxn}=\sum [n\times \Delta G^o_{(product)}]-\sum [n\times \Delta G^o_{(reactant)}]](/tpl/images/0536/5267/f0852.png)
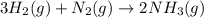
![\Delta G^o_{rxn}=[(2\times \Delta G^o_{(NH_3(g))})]-[(1\times \Delta G^o_{(N_2)})+(3\times \Delta G^o_{(H_2)})]](/tpl/images/0536/5267/7de49.png)
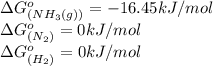
![\Delta G^o_{rxn}=[(2\times (-16.45))]-[(1\times (0))+(3\times (0))]\\\\\Delta G^o_{rxn}=-32.9kJ/mol](/tpl/images/0536/5267/8bdb6.png)
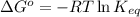
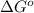 = standard Gibbs free energy = -32.9 kJ/mol = -35900 J/mol (Conversion factor: 1 kJ = 1000 J )
= standard Gibbs free energy = -32.9 kJ/mol = -35900 J/mol (Conversion factor: 1 kJ = 1000 J )![25^oC=[273+25]K=298K](/tpl/images/0536/5267/0e82f.png)
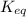 = equilibrium constant at 25°C = ?
= equilibrium constant at 25°C = ?