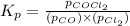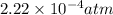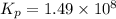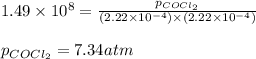Chemistry, 07.03.2020 00:09 youngbeauty17
The Kp for the reaction below is 1.49 × 108 at 100.0°C:CO(g) + Cl2(g) → COCl2(g)In an equilibrium mixture of the three gases, PCO = PCl2 = 2.22 × 10-4 atm. The partial pressure of the product, phosgene (COCl2), is atm.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 07:00
This image is an example of a(n) a) atom. b) compound. c) mixture. d) molecule.
Answers: 1

You know the right answer?
The Kp for the reaction below is 1.49 × 108 at 100.0°C:CO(g) + Cl2(g) → COCl2(g)In an equilibrium mi...
Questions

Computers and Technology, 14.06.2021 15:20














Computers and Technology, 14.06.2021 15:20



Mathematics, 14.06.2021 15:20


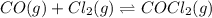
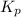 for above equation follows:
for above equation follows: