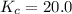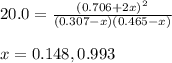Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
The p sub shell can hold up to 8 electrons in an atom. true or false?
Answers: 1

Chemistry, 22.06.2019 06:30
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3

Chemistry, 22.06.2019 08:00
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

You know the right answer?
A mixture of 0.307 M Cl 2 , 0.465 M F 2 , and 0.706 M ClF is enclosed in a vessel and heated to 2500...
Questions

Computers and Technology, 20.03.2020 03:27

Chemistry, 20.03.2020 03:27

Social Studies, 20.03.2020 03:27




Mathematics, 20.03.2020 03:27





English, 20.03.2020 03:28

Mathematics, 20.03.2020 03:28




Mathematics, 20.03.2020 03:28



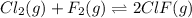
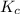 for above equation follows:
for above equation follows:![K_c=\frac{[ClF]^2}{[Cl_2][F_2]}](/tpl/images/0536/8605/4a1ee.png)