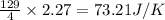Chemistry, 07.03.2020 00:46 flowerrbabie
Consider the reaction 4HCl(g) + O2(g)2H2O(g) + 2Cl2(g) Using standard thermodynamic data at 298K, calculate the entropy change for the surroundings when 2.27 moles of HCl(g) react at standard conditions. Ssurroundings = J/K

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which is true of the reactants in this displacement reaction? fe + 2hcl fecl2 + h2 a. the reactants are located to the left of the arrow in the chemical equation. b. the reactants contain 1 iron atom, 2 hydrogen atoms, and 1 chlorine atom. c. the reactants are the atoms, molecules, or compounds formed in the reaction. d. the reactants have the same physical and chemical properties as the products.
Answers: 1



Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1
You know the right answer?
Consider the reaction 4HCl(g) + O2(g)2H2O(g) + 2Cl2(g) Using standard thermodynamic data at 298K, ca...
Questions


English, 20.09.2019 05:30






History, 20.09.2019 05:30

Mathematics, 20.09.2019 05:30


History, 20.09.2019 05:30


Health, 20.09.2019 05:30

Mathematics, 20.09.2019 05:30






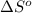 for the surrounding when given amount of HCl gas is reacted is 73.21 J/K
for the surrounding when given amount of HCl gas is reacted is 73.21 J/K![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0536/8507/52737.png)
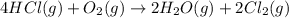
![\Delta S^o_{rxn}=[(2\times \Delta S^o_{(Cl_2(g))})+(2\times \Delta S^o_{(H_2O(g))})]-[(4\times \Delta S^o_{(HCl(g))})+(1\times \Delta S^o_{(O_2(g))})]](/tpl/images/0536/8507/2475f.png)
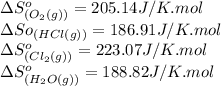
![\Delta S^o_{rxn}=[(2\times (223.07))+(2\times (188.82))]-[(4\times (186.91))+(1\times (205.14))]\\\\\Delta S^o_{rxn}=-129J/K](/tpl/images/0536/8507/8e27e.png)