Chemistry, 07.03.2020 01:42 ellaemtagedeane
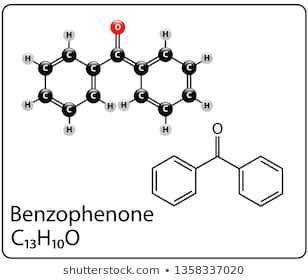
Benzene was used in the following multistep reaction to produce a product with formula C13H10O. Use the synthetic scheme and the mass spectrum provided to draw the structure of the final product.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
Benzene was used in the following multistep reaction to produce a product with formula C13H10O. Use...
Questions

Mathematics, 27.07.2019 11:40



Mathematics, 27.07.2019 11:40




Mathematics, 27.07.2019 11:40

History, 27.07.2019 11:40



Mathematics, 27.07.2019 11:40






Chemistry, 27.07.2019 11:40


Mathematics, 27.07.2019 11:40