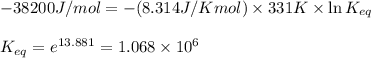Chemistry, 07.03.2020 03:46 Joshuafranklindude
In the Mond process for the purification of nickel, carbon monoxide is reacted with heated nickel to produce Ni(CO)4, which is a gas and can therefore be separated from solid impurities: Ni(s) + 4CO(g) ⇌ Ni(CO)4(g) Given that the standard free energies of formation of CO(g) and Ni(CO)4(g) are −137.3 and −587.4 kJ/mol, respectively, calculate the equilibrium constant of the reaction at 58.0°C. Assume that ΔG o f is temperature-independent.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2
You know the right answer?
In the Mond process for the purification of nickel, carbon monoxide is reacted with heated nickel to...
Questions



Chemistry, 03.08.2019 03:00




Mathematics, 03.08.2019 03:00


Biology, 03.08.2019 03:00


Social Studies, 03.08.2019 03:00

Physics, 03.08.2019 03:00





Mathematics, 03.08.2019 03:00

Advanced Placement (AP), 03.08.2019 03:00

History, 03.08.2019 03:00

Mathematics, 03.08.2019 03:00

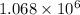
![\Delta G^o_{rxn}=\sum [n\times \Delta G^o_{(product)}]-\sum [n\times \Delta G^o_{(reactant)}]](/tpl/images/0537/4791/f0852.png)
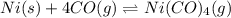
![\Delta G^o_{rxn}=[(1\times \Delta G^o_{(Ni(CO)_4(g))})]-[(1\times \Delta G^o_{(Ni(s))})+(4\times \Delta G^o_{(CO(g))})]](/tpl/images/0537/4791/5f698.png)

![\Delta G^o_{rxn}=[(1\times (-587.4))]-[(1\times (0))+(4\times (-137.3))]\\\\\Delta G^o_{rxn}=-38.2kJ/mol](/tpl/images/0537/4791/37589.png)

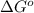 = Standard Gibbs free energy = -38.2 kJ/mol = -38200 J/mol (Conversion factor: 1 kJ = 1000 J )
= Standard Gibbs free energy = -38.2 kJ/mol = -38200 J/mol (Conversion factor: 1 kJ = 1000 J )![58^oC=[273+58]K=331K](/tpl/images/0537/4791/2a33f.png)
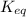 = equilibrium constant at 58°C = ?
= equilibrium constant at 58°C = ?