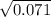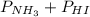Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1

Chemistry, 23.06.2019 01:30
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1

Chemistry, 23.06.2019 13:50
Which compounds are the brønsted-lowry bases in this equilibrium: hc2o4– + h2bo3– h3bo3 + c2o42– ? a. h2bo3– and h3bo3 b. hc2o4– and h3bo3 c. hc2o4– and c2o42– d. h2bo3– and c2o42– e. hc2o4– and h2bo3–
Answers: 2

Chemistry, 24.06.2019 00:30
Nitric acid is often manufactured from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming of natural gas, in a two-step process. in the first step, nitrogen and hydrogen react to form ammonia:
Answers: 2
You know the right answer?
If Kp For This Reaction Is 0.071 At 100 °C (when The Partial Pressures Are Measured In Atmospheres),...
Questions

Mathematics, 24.09.2019 23:20



History, 24.09.2019 23:20

History, 24.09.2019 23:20


Social Studies, 24.09.2019 23:20


English, 24.09.2019 23:20


Mathematics, 24.09.2019 23:20

English, 24.09.2019 23:20

Mathematics, 24.09.2019 23:20







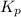 = 0.071 at 100°C
= 0.071 at 100°C![[P_{NH_3}][P_{HI}]](/tpl/images/0537/6634/19fd4.png)
![[x][x]](/tpl/images/0537/6634/ce5d5.png)