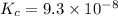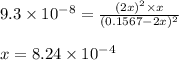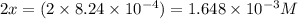Chemistry, 07.03.2020 04:30 liamgreene90
2 H2S(g) ⇄ 2 H2(g) + S2(g) Kc = 9.3× 10-8 at 400ºC 0.47 moles of H2S are placed in a 3.0 L container and the system is allowed to reach equilibrium. Calculate the concentration of H2 at equilibrium.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
2 H2S(g) ⇄ 2 H2(g) + S2(g) Kc = 9.3× 10-8 at 400ºC 0.47 moles of H2S are placed in a 3.0 L container...
Questions

Mathematics, 04.09.2019 02:30

Computers and Technology, 04.09.2019 02:30








History, 04.09.2019 02:30


Mathematics, 04.09.2019 02:30


Mathematics, 04.09.2019 02:30


Mathematics, 04.09.2019 02:30


Physics, 04.09.2019 02:30

Mathematics, 04.09.2019 02:30

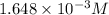
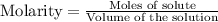
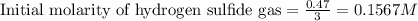
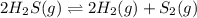
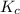 for above equation follows:
for above equation follows:![K_c=\frac{[H_2]^2[S_2]}{[H_2S]^2}](/tpl/images/0537/6756/3ac5e.png)