
Chemistry, 07.03.2020 04:38 mihirkantighosh
For the reaction Fe2O3(s) + 3H2(g)2Fe(s) + 3H2O(g) H°rxn = 98.8 kJ and S°rxn = 142.5 J/K The standard free energy change for the reaction of 2.47 moles of Fe2O3(s) at 267 K, 1 atm would be kJ. This reaction is (reactant, product) favored under standard conditions at 267 K. Assume that H°rxn and S°rxn are independent of temperature.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 23:30
Rank the following four acids in order of increasing bronsted acidity : h2f+ , ch3oh, (ch3)2oh+ , ch3sh2+
Answers: 3

Chemistry, 23.06.2019 01:00
Which polymers are most closely related? a. protein and nucleic acids b. cellulose and starch c. nucleic acids and starch d. nucleic acids and cellulose
Answers: 2

Chemistry, 23.06.2019 01:10
A5.00 g of a in . g of at aa 5.00 g of b in . g of .?at .
Answers: 1
You know the right answer?
For the reaction Fe2O3(s) + 3H2(g)2Fe(s) + 3H2O(g) H°rxn = 98.8 kJ and S°rxn = 142.5 J/K The standar...
Questions

History, 18.07.2019 20:00

Geography, 18.07.2019 20:00

Mathematics, 18.07.2019 20:00





Mathematics, 18.07.2019 20:00

Mathematics, 18.07.2019 20:00



Physics, 18.07.2019 20:00


Mathematics, 18.07.2019 20:00

Mathematics, 18.07.2019 20:00

Mathematics, 18.07.2019 20:00


Mathematics, 18.07.2019 20:00

Biology, 18.07.2019 20:00

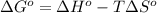
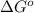 = standard Gibbs free energy = ?
= standard Gibbs free energy = ?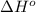 = standard enthalpy = 98.8 kJ = 98800 J
= standard enthalpy = 98.8 kJ = 98800 J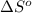 = standard entropy = 142.5 J/K
= standard entropy = 142.5 J/K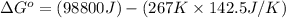
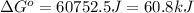
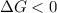 and reaction will be favored in the forward direction that means favored in products.A reaction to be non-spontaneous when
and reaction will be favored in the forward direction that means favored in products.A reaction to be non-spontaneous when 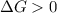 and reaction will be favored in the backward direction that means favored in reactants.
and reaction will be favored in the backward direction that means favored in reactants.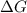 is more than zero that means the reaction is non-spontaneous and reaction will be favored in the backward direction that means favored in reactants.
is more than zero that means the reaction is non-spontaneous and reaction will be favored in the backward direction that means favored in reactants.

