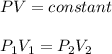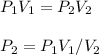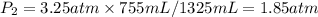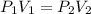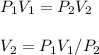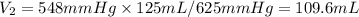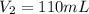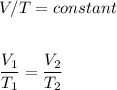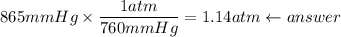Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2



You know the right answer?
I need help with 1,2,3, and 4
...
...
Questions

Mathematics, 12.05.2021 16:40

Mathematics, 12.05.2021 16:40

Geography, 12.05.2021 16:40



Mathematics, 12.05.2021 16:40




Mathematics, 12.05.2021 16:40

Business, 12.05.2021 16:40



Mathematics, 12.05.2021 16:40


Arts, 12.05.2021 16:40

Mathematics, 12.05.2021 16:40


Mathematics, 12.05.2021 16:40