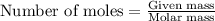Problem Page Question An analytical chemist weighs out of an unknown monoprotic acid into a volumetric flask and dilutes to the mark with distilled water. He then titrated this solution with solution. When the titration reaches the equivalence point, the chemist finds he has added of solution. Calculate the molar mass of the unknown acid. Round your answer to significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2


Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1
You know the right answer?
Problem Page Question An analytical chemist weighs out of an unknown monoprotic acid into a volumetr...
Questions



Mathematics, 16.05.2021 20:10

Mathematics, 16.05.2021 20:10

Mathematics, 16.05.2021 20:10






Mathematics, 16.05.2021 20:10

Social Studies, 16.05.2021 20:10


Mathematics, 16.05.2021 20:10

Social Studies, 16.05.2021 20:10

Mathematics, 16.05.2021 20:10

English, 16.05.2021 20:10

Mathematics, 16.05.2021 20:10

Social Studies, 16.05.2021 20:10

Mathematics, 16.05.2021 20:10

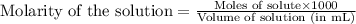
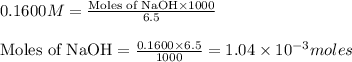

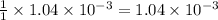 moles of HA
moles of HA