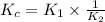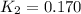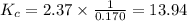Chemistry, 10.03.2020 01:13 tiffany991223
Two reactions and their equilibrium constants are given. A + 2 B − ⇀ ↽ − 2 C K 1 = 2.37 2 C − ⇀ ↽ − D K 2 = 0.170 Calculate the value of the equilibrium constant for the reaction D − ⇀ ↽ − A + 2 B .

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2



Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1
You know the right answer?
Two reactions and their equilibrium constants are given. A + 2 B − ⇀ ↽ − 2 C K 1 = 2.37 2 C − ⇀ ↽ −...
Questions




Mathematics, 27.02.2020 22:50

Mathematics, 27.02.2020 22:50






History, 27.02.2020 22:50

Mathematics, 27.02.2020 22:50







Arts, 27.02.2020 22:50

History, 27.02.2020 22:50

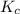 for the net reaction is 13.94
for the net reaction is 13.94![A+2B\xrightarrow[]{K_1} 2C](/tpl/images/0539/5006/bf2d5.png)
![2C\xrightarrow[]{K_2} D](/tpl/images/0539/5006/8564b.png)
![D\xrightarrow[]{K_c} A+2B](/tpl/images/0539/5006/86185.png)