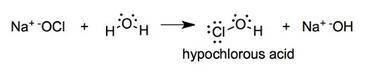
Once the ionic solid has dissolved, the anion that is formed is able to react as a base, with water as the acid. Write the net acid-base reaction that occurs when dissolved NaClO reacts with water. (Use the lowest possible coefficients. Omit states-of-matter in your answer.)

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 23.06.2019 10:30
Silver is a white metal that is an excellent conductor. silver tarnishes when exposed to air and light. the density of silver is 10.49 g/cm3. the melting point is 962oc and the boiling point is 2000oc. a chemical property of silver is
Answers: 3

Chemistry, 23.06.2019 17:10
Two changes are described below. a green banana turns yellow and ripens. a layer of rust forms on an iron nail. which statement is true about the two changes? a) both are chemical changes because new substances are formed. b) both are physical changes because only the physical state of the substances change. c) a is a physical change due to a change of state, but b is a chemical change because new molecules are formed. d) a is a chemical change due to a change of state, but b is a physical change because new molecules are formed.
Answers: 1
You know the right answer?
Once the ionic solid has dissolved, the anion that is formed is able to react as a base, with water...
Questions

Mathematics, 05.05.2020 02:17


Mathematics, 05.05.2020 02:17

Biology, 05.05.2020 02:17

Mathematics, 05.05.2020 02:17

Mathematics, 05.05.2020 02:17


Mathematics, 05.05.2020 02:17


Mathematics, 05.05.2020 02:17

Mathematics, 05.05.2020 02:17

Mathematics, 05.05.2020 02:17

Mathematics, 05.05.2020 02:17





Biology, 05.05.2020 02:17