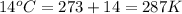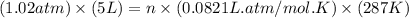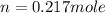Chemistry, 10.03.2020 20:35 mwilliams457
Calculate the amount of oxygen gas collected by the displacement of water at 14◦C if the atmospheric pressure is 790 Torr and the volume is 5 L. The vapor pressure of water at 14◦C is 12 Torr. 1. 0.217 mol 2. 0.00335 mol 3. 0.0184 mol 4. 4.46 mol 5. 0.224 mol

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which of the following elements is a representative element? a. chromium (cr) b. aluminum (al) c. mercury (hg) d. silver (ag)
Answers: 3

Chemistry, 22.06.2019 03:10
Between 2014 and 2016, more than 25,000 children in flint, michigan, drank water that was contaminated with lead from lead pipes. during this time, the city claimed the water was safe to drink. which of these actions could the city have taken to ensure that the drinking water was free from lead?
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2
You know the right answer?
Calculate the amount of oxygen gas collected by the displacement of water at 14◦C if the atmospheric...
Questions

English, 22.01.2021 03:00


Mathematics, 22.01.2021 03:00






Mathematics, 22.01.2021 03:00


Spanish, 22.01.2021 03:00



Arts, 22.01.2021 03:00


Mathematics, 22.01.2021 03:00




Mathematics, 22.01.2021 03:00

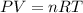
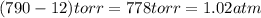 (1 atm = 760 torr)
(1 atm = 760 torr)