
Consider the following mechanism for the oxidation of bromide ions by hydrogen peroxide in aqueous acid solution. H+ + H2O2 ? H3O2+ (rapid equilibrium) H3O2+ + Br- ? HOBr + H2O (slow) HOBr+H+ +Br- ?Br2 +H2O(fast) Which rate law is consistent with this mechanism?
a. k[Br-][H+]-1[H2O2]-1
b. k[H+][H2O2][Br-]
c. k[H+][H2O2]
d. k[HOBr][H+][Br-]

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3


Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

You know the right answer?
Consider the following mechanism for the oxidation of bromide ions by hydrogen peroxide in aqueous a...
Questions

English, 08.10.2019 16:30

Computers and Technology, 08.10.2019 16:30


Mathematics, 08.10.2019 16:30















Computers and Technology, 08.10.2019 16:30

![\text{Rate}=k'[H+][H_2O_2][Br^-]](/tpl/images/0541/5354/e35f3.png)
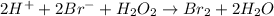
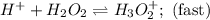
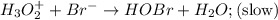
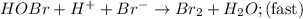
![\text{Rate}=k[H_3O_2^+][Br^-]](/tpl/images/0541/5354/37d2f.png) ......(1)
......(1)![[H_3O_2^+]](/tpl/images/0541/5354/85c88.png) is not appearing as a reactant in the overall reaction. So, we apply steady state approximation in it.
is not appearing as a reactant in the overall reaction. So, we apply steady state approximation in it.![K=\frac{[H_3O_2^+]}{[H^+][H_2O_2]}](/tpl/images/0541/5354/4cd3a.png)
![[H_3O_2^+]=K[H^+][H_2O_2]](/tpl/images/0541/5354/15cd9.png)
![\text{Rate}=k.K[H^+][H_2O_2][Br^-]\\\\\text{Rate}=k'[H+][H_2O_2][Br^-]](/tpl/images/0541/5354/f918c.png)


