
Chemistry, 11.03.2020 06:06 QuestionsAnsweredNow
A mixture of CO2 and Kr weighs 41.0 g and exerts a pressure of 0.729 atm in its container. Since Kr is expensive, you wish to recover it from the mixture. After the CO2 is completely removed by absorption with NaOH(s), the pressure in the container is 0.193 atm. (a) How many grams of CO2 were originally present? (b) How many grams of Kr can you recover?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3
You know the right answer?
A mixture of CO2 and Kr weighs 41.0 g and exerts a pressure of 0.729 atm in its container. Since Kr...
Questions

Mathematics, 02.07.2019 12:00



Mathematics, 02.07.2019 12:00


Chemistry, 02.07.2019 12:00


Biology, 02.07.2019 12:00



Mathematics, 02.07.2019 12:00


Social Studies, 02.07.2019 12:00


History, 02.07.2019 12:00

Physics, 02.07.2019 12:00

Mathematics, 02.07.2019 12:00

History, 02.07.2019 12:00


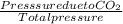
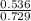
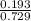
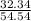 * 100 %
* 100 % 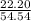 * 100 %
* 100 % 

