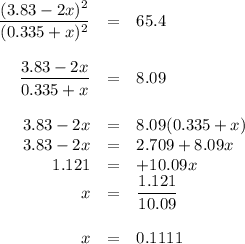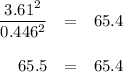Chemistry, 11.03.2020 22:26 gshreya2005
At 500 °C, hydrogen iodide decomposes according to 2 HI ( g ) − ⇀ ↽ − H 2 ( g ) + I 2 ( g ) For HI ( g ) heated to 500 °C in a 1.00 L reaction vessel, chemical analysis determined these concentrations at equilibrium: [ H 2 ] = 0.335 M , [ I 2 ] = 0.335 M , and [ HI ] = 2.83 M . If an additional 1.00 mol of HI ( g ) is introduced into the reaction vessel, what are the equilibrium concentrations after the new equilibrium has been reached?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2
You know the right answer?
At 500 °C, hydrogen iodide decomposes according to 2 HI ( g ) − ⇀ ↽ − H 2 ( g ) + I 2 ( g ) For HI (...
Questions



Computers and Technology, 14.11.2021 09:50


Computers and Technology, 14.11.2021 14:00


Mathematics, 14.11.2021 14:00




Mathematics, 14.11.2021 14:00


SAT, 14.11.2021 14:00


Chemistry, 14.11.2021 14:00

History, 14.11.2021 14:00

History, 14.11.2021 14:00



![K_{\text{eq}} = \dfrac{\text{[HI]$^{2}$}}{\text{[H$_{2}]$[I$_{2}$]}} = \dfrac{2.83^{2}}{0.335 \times 0.335} = 65.4](/tpl/images/0543/5148/5b908.png)
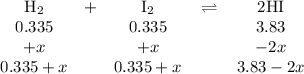
![K_{\text{c}} = \dfrac{\text{[HI]$^{2}$}}{\text{[H$_{2}$][I$_2$]}} = \dfrac{(3.83 - 2x)^{2}}{(0.335 + x)^{2}} = 65.4](/tpl/images/0543/5148/b1d26.png)