
Chemistry, 11.03.2020 22:56 amberpetty4288
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that goes with this equilibrium constant. (Use H3O+ instead of H+.)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1


Chemistry, 23.06.2019 13:30
The two isotopes of chlorine are 3517cl and 3717cl. which isotope is the most abundant?
Answers: 1
You know the right answer?
The value of Ka for acetic acid , CH3COOH , is 1.80×10-5 . Write the equation for the reaction that...
Questions

Mathematics, 23.07.2019 17:00


Mathematics, 23.07.2019 17:00

Mathematics, 23.07.2019 17:00



History, 23.07.2019 17:00

History, 23.07.2019 17:00


History, 23.07.2019 17:00





History, 23.07.2019 17:00

Biology, 23.07.2019 17:00

Social Studies, 23.07.2019 17:00


Mathematics, 23.07.2019 17:00

English, 23.07.2019 17:00

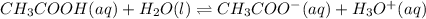
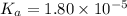
![K_c=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH][H_2O]}](/tpl/images/0543/5879/7c275.png)
![K_a=K_c\times [H_2O]=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/125a5.png)
![[H_2O]=1](/tpl/images/0543/5879/b8579.png)
![K_a=\frac{[CH_3COO^-][H_3O^+]}{[CH-3COOH]}](/tpl/images/0543/5879/e4df0.png)


