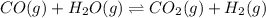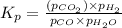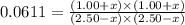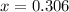Chemistry, 12.03.2020 00:02 Mitchmorgan3816
CO(g ) + H2O(g ) <---> CO2(g ) + H2(g ), Kc = 0.0611 at 2000 K . A reaction mixture initially contains a CO partial pressure of 2.50 atm, an H2O partial pressure of 2.50 atm, a CO2 partial pressure of 1.00 atm, and an H2 partial pressure of 1.00 atm at 2000 K. Calculate the equilibrium partial pressure of CO

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Measuring which physical property is most likely to produce the most precise results when trying to identify a substance
Answers: 1

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1


Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1
You know the right answer?
CO(g ) + H2O(g ) <---> CO2(g ) + H2(g ), Kc = 0.0611 at 2000 K . A reaction mixture initially...
Questions

Mathematics, 23.11.2020 17:30

Social Studies, 23.11.2020 17:30


English, 23.11.2020 17:30

History, 23.11.2020 17:30



Mathematics, 23.11.2020 17:40

Physics, 23.11.2020 17:40


English, 23.11.2020 17:40

Mathematics, 23.11.2020 17:40

Mathematics, 23.11.2020 17:40

Mathematics, 23.11.2020 17:40


Mathematics, 23.11.2020 17:40



Mathematics, 23.11.2020 17:40

Mathematics, 23.11.2020 17:40

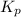 is the constant of a certain reaction at equilibrium.
is the constant of a certain reaction at equilibrium.