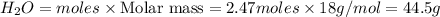Chemistry, 13.03.2020 03:38 victoriapellam04
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many modern chemical processes. In the first step of the Ostwald process, ammonia is reacted with oxygen gas to produce nitric oxide and water. What is the maximum mass of H 2 O that can be produced by combining 65.8 g of each reactant? 4 NH 3 ( g ) + 5 O 2 ( g ) ⟶ 4 NO ( g ) + 6 H 2 O ( g )

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:10
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1


Chemistry, 23.06.2019 06:30
Which of these natural resources is non-renewable a.corn b.wind c.geothermal d.natural gas
Answers: 2
You know the right answer?
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many mod...
Questions

Mathematics, 22.01.2021 20:20

Mathematics, 22.01.2021 20:20

Chemistry, 22.01.2021 20:20




Mathematics, 22.01.2021 20:20





Mathematics, 22.01.2021 20:20

English, 22.01.2021 20:20






Mathematics, 22.01.2021 20:20

Mathematics, 22.01.2021 20:20

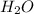 will be produced from the given masses of both reactants.
will be produced from the given masses of both reactants.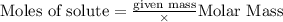
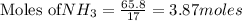
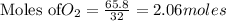
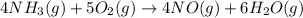
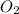 require 4 moles of
require 4 moles of 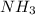
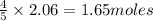 of
of 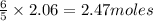 of
of