PLZ HELP, GIVING BRAINLIEST!!
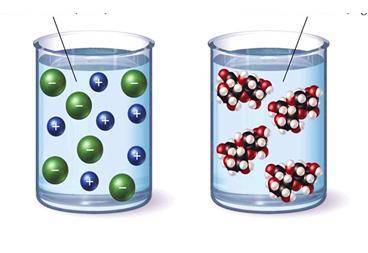
In class, students were given the pictures below and asked to pi...

Chemistry, 14.03.2020 00:29 cxttiemsp021
PLZ HELP, GIVING BRAINLIEST!!
In class, students were given the pictures below and asked to pick a solution that would conduct electricity and to justify their choice.
Based on the model above, which student's argument is correct?
A. Student B claims that the left beaker contains a covalent compound because the solute breaks apart into charged particles.
B. Student D claims to identify the solute as either ionic or covalent more information is needed than what is provided in the model.
C. Student C claims that the right beaker contains an ionic compound because the solute stays together when dissolved.
D. Student A claims that the left beaker contains an ionic compound because the solute breaks apart into charged particles.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3


Chemistry, 23.06.2019 00:00
How is the way a mixture is combined different from how a compound is combined?
Answers: 3
You know the right answer?
Questions


Physics, 08.04.2020 01:30






Mathematics, 08.04.2020 01:30



History, 08.04.2020 01:30











