Chemistry, 14.03.2020 06:55 rosemarybooker
The following reaction is spontaneous as written when the components are in their standard states:
3 Zn(s) +2 Cr3+(aq) →3 Zn2+(aq) +2 Cr(s)
If the [Zn2+] is 4 molL−1, determine the value of [Cr3+] below which the reaction will be spontaneous in the opposite direction.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3


Chemistry, 23.06.2019 01:30
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2
You know the right answer?
The following reaction is spontaneous as written when the components are in their standard states:
Questions

Health, 25.09.2020 07:01



History, 25.09.2020 07:01

Physics, 25.09.2020 07:01

English, 25.09.2020 07:01


Chemistry, 25.09.2020 07:01


Chemistry, 25.09.2020 07:01



History, 25.09.2020 07:01

Mathematics, 25.09.2020 07:01




Mathematics, 25.09.2020 07:01

History, 25.09.2020 07:01

Geography, 25.09.2020 07:01

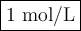
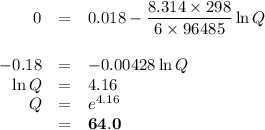
![\begin{array}{rcl}Q & = & \dfrac{\text{[Zn$^{2+}$]$^{3}$}}{\text{[Cr}^{3+}]^{2}}\\\\64.0 & = & \dfrac{4^{3}}{\text{[Cr}^{3+}]^{2}}\\\\\text{[Cr}^{3+}]^{2}& = & \dfrac{64}{64.0}\\\\& = & 1\\\text{[Cr}^{3+}] & = & \textbf{1 mol/L}\\\end{array}\\\text{[Cr$^{3+}$] must be less than $\large \boxed{\textbf{1 mol/L}}$ for the reaction to be spontaneous in the reverse}\\\text{direction.}](/tpl/images/0547/6685/5fcfe.png)