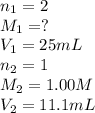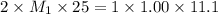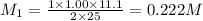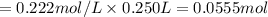Chemistry, 16.03.2020 17:02 staxeeyy767
A 5.00g quantity of a diprotic acid was dissolved in water and made up exactly 250 mL. Calculate the molar mass if the acid is 25.0 mL of this solution required 11.1 mL of 1.00 KOH for neutralization. Assume both protons of the acid were titrated.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 13:40
Can someone me with 6 to 10 plz this is for masteries test.
Answers: 1

You know the right answer?
A 5.00g quantity of a diprotic acid was dissolved in water and made up exactly 250 mL. Calculate the...
Questions


Chemistry, 04.01.2021 20:30






Computers and Technology, 04.01.2021 20:30


Social Studies, 04.01.2021 20:30


Mathematics, 04.01.2021 20:30



Biology, 04.01.2021 20:30

Mathematics, 04.01.2021 20:30




Mathematics, 04.01.2021 20:30

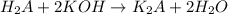
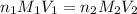 ( neutralization )
( neutralization )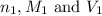 are the n-factor, molarity and volume of diprotic acid
are the n-factor, molarity and volume of diprotic acid 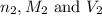 are the n-factor, molarity and volume of base which is KOH.
are the n-factor, molarity and volume of base which is KOH.