
Chemistry, 16.03.2020 18:59 nicollexo21
You are instructed to create 900. mL of a 0.29 M phosphate buffer with a pH of 7.8. You have phosphoric acid and the sodium salts NaH2PO4, Na2HPO4, and Na3PO4 available. (Enter all numerical answers to three significant figures.)
H3PO4(s) + H2O(l) <> H3O+(aq) + H2PO4−(aq) Ka1 = 6.9 ✕ 10−3
H2PO4−(aq) + H2O(l) <> H3O+(aq) + HPO42−(aq) Ka2 = 6.2 ✕ 10−8
HPO42−(aq) + H2O(l) <> H3O+(aq) + PO43−(aq) Ka3 = 4.8 ✕ 10−13
Which of the available chemicals will you use for the acid component of your buffer?
A. H3PO4
B. NaH2PO4
C. Na2HPO4
D. Na3PO4
Which of the available chemicals will you use for the base component of your buffer?
A. H3PO4B. NaH2PO4C. Na2HPO4D. Na3PO4

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

Chemistry, 22.06.2019 21:20
Juju.01) 5 geologic events a group of students designed an experiment in an ice rink to represent the solar system. the steps of the experiment are listed below. 5: geologic events 1. choose a student to represent the sun. the planets are represented by two tennis balls. 2. ask the student to hold the tennis balls in each palm and spin on the ice with arms stretched out. 3. ask the student to draw in the arms after about 10 spins. 4. observe the student's arms rotate faster when they are closer to the body. 05 geologic events enors) the experiment most likely demonstrates that (2 points) 07 discussion-based sessment/module planets exert gravitational force on the sun the sun exerts gravitational force on the planets 3.07 discussion-based ssessment speed of a planet depends on its distance from the sun new version available! (3.0.119) get it now submit 18.07: module exam description
Answers: 3
You know the right answer?
You are instructed to create 900. mL of a 0.29 M phosphate buffer with a pH of 7.8. You have phospho...
Questions




Mathematics, 16.10.2021 19:50


Mathematics, 16.10.2021 20:00


Mathematics, 16.10.2021 20:00



Computers and Technology, 16.10.2021 20:00

Mathematics, 16.10.2021 20:00



SAT, 16.10.2021 20:00

Biology, 16.10.2021 20:00


Mathematics, 16.10.2021 20:00


Social Studies, 16.10.2021 20:00

![pKa_1=-Log[6.9x10^-^3]=2.16](/tpl/images/0548/8384/6a482.png)
![pKa_2=-Log[6.2x10^-^8]=7.20](/tpl/images/0548/8384/8337b.png)
![pKa_3=-Log[4.8x10^-^13]=12.31](/tpl/images/0548/8384/78632.png)
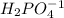 is the acid and
is the acid and 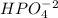 is the base. Therefore the answer for the first question is B and the answer for the second question is C.
is the base. Therefore the answer for the first question is B and the answer for the second question is C.

