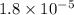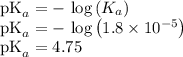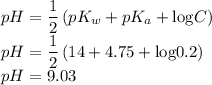Chemistry, 16.03.2020 20:24 arionaking59p71cfc
Calculate the pH of KC2H3O2 solution. Ka for HC2H3O2 is 1.8x10-5

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 23.06.2019 07:20
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3
You know the right answer?
Calculate the pH of KC2H3O2 solution. Ka for HC2H3O2 is 1.8x10-5...
Questions

Mathematics, 21.02.2021 18:50




Biology, 21.02.2021 18:50

Mathematics, 21.02.2021 18:50

Chemistry, 21.02.2021 19:00



History, 21.02.2021 19:00

Mathematics, 21.02.2021 19:00

Mathematics, 21.02.2021 19:00



English, 21.02.2021 19:00





Mathematics, 21.02.2021 19:00

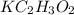 is a salt of a weak acid acetic acid and a strong base KOH. This salt will hydrolyze in water to give an alkaline solution.
is a salt of a weak acid acetic acid and a strong base KOH. This salt will hydrolyze in water to give an alkaline solution.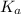 of acetic acid =
of acetic acid =