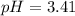Chemistry, 17.03.2020 00:57 claytonhopkins
A 1.50 L buffer solution is 0.250 M in HF and 0.250 M in NaF. Calculate the pH of the solution after the addition of 0.100 moles of solid NaOH. Assume no volume change upon the addition of base. The Ka for HF is 6.8 × 10-4.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:50
Which term refers to a property that depends only on the amount of a substance? ©@
Answers: 2

Chemistry, 21.06.2019 22:00
Afamily is one another name for a group on the table of elements.
Answers: 1

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3
You know the right answer?
A 1.50 L buffer solution is 0.250 M in HF and 0.250 M in NaF. Calculate the pH of the solution after...
Questions


Mathematics, 11.02.2021 17:50

Mathematics, 11.02.2021 17:50

Mathematics, 11.02.2021 17:50




Mathematics, 11.02.2021 17:50

Mathematics, 11.02.2021 17:50







Mathematics, 11.02.2021 17:50



Mathematics, 11.02.2021 17:50

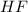 .
.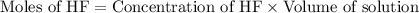
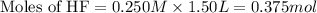
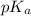 .
.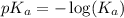
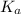 in this expression, we get:
in this expression, we get: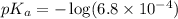
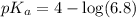
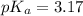
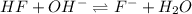
![pH=pK_a+\log \frac{[Salt]}{[Acid]}](/tpl/images/0549/7799/e961a.png)
![pH=pK_a+\log \frac{[F^-]}{[HF]}](/tpl/images/0549/7799/bef2f.png)
![pH=3.17+\log [\frac{(\frac{0.475}{1.50})}{(\frac{0.275}{1.50})}]](/tpl/images/0549/7799/a4ae7.png)