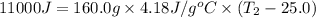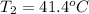Consider the dissolution of CaCl₂: CaCl₂(s) → Ca²⁺(aq) + 2Cl⁻(aq) ∆H = -81.5 kJ/mol A 15.0-g sample of CaCl₂ is dissolved in 145 g water with both substances at 25.0°C. Calculate the final temperature of the solution assuming no heat loss to the surroundings and assuming the solution has a specific heat capacity of 4.18 J/°C・g.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Type the letter that represents the correct location for each particle type below.
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
Consider the dissolution of CaCl₂: CaCl₂(s) → Ca²⁺(aq) + 2Cl⁻(aq) ∆H = -81.5 kJ/mol A 15.0-g sample...
Questions

Mathematics, 30.01.2021 01:40

History, 30.01.2021 01:40

Mathematics, 30.01.2021 01:40

English, 30.01.2021 01:40




Mathematics, 30.01.2021 01:40


English, 30.01.2021 01:40


Mathematics, 30.01.2021 01:40


Biology, 30.01.2021 01:40




Mathematics, 30.01.2021 01:40


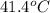
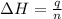
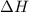 = enthalpy change = 81.5 kJ/mol
= enthalpy change = 81.5 kJ/mol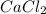 = 15.0 g
= 15.0 g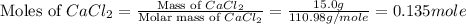
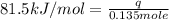
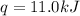
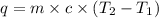
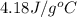
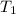 = initial temperature =
= initial temperature = 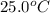
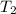 = final temperature = ?
= final temperature = ?