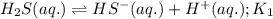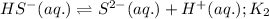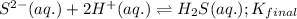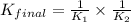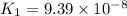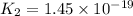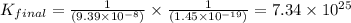Chemistry, 17.03.2020 04:33 channarlawassociate
Given the two reactions H2S(aq)⇌HS−(aq)+H+(aq), H2S(aq)⇌HS−(aq)+H+(aq), K1K1K_1 = 9.39×10−8, and HS−(aq)⇌S2−(aq)+H+(aq), HS−(aq)⇌S2−(aq)+H+(aq), K2K2K_2 = 1.45×10−19, what is the equilibrium constant KfinalKfinalK_final for the following reaction? S2−(aq)+2H+(aq)⇌H2S(aq)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 23.06.2019 00:30
Ok, so i have 2 questions. try to answer them both: (the topic is fire) 1) how can you represent the chemical reaction of fire? 2) what kind of bond is formed in this chemical reaction
Answers: 3
You know the right answer?
Given the two reactions H2S(aq)⇌HS−(aq)+H+(aq), H2S(aq)⇌HS−(aq)+H+(aq), K1K1K_1 = 9.39×10−8, and HS−...
Questions



Computers and Technology, 16.10.2020 14:01


History, 16.10.2020 14:01

Health, 16.10.2020 14:01

English, 16.10.2020 14:01

Mathematics, 16.10.2020 14:01

Social Studies, 16.10.2020 14:01





Mathematics, 16.10.2020 14:01

Mathematics, 16.10.2020 14:01



Advanced Placement (AP), 16.10.2020 14:01


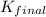 for the net reaction is
for the net reaction is