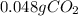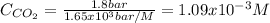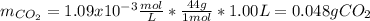Chemistry, 17.03.2020 05:24 adrian128383
How many grams of CO2 are dissolved in a 1.00 L bottle of carbonated water at 298 K if the pressure used in the carbonation process was 1.8 bar? The density of water at this temperature is 998 kg⋅m−3. The Henry's law constant for aqueous solution of CO2 at this temperature is 1.65×103bar.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Imagine that you own a property that is exactly 2.2 acres large. you want to sell your property, but your realtor tells you that you cannot sell your land by the acre. in order to sell your land you need to determine the area you own in units of square meters? given that there are 1.6 kilometers in 1 mile and 640 acres in 1 square mile, what is the area of land that you own in square meters square meters?
Answers: 2

Chemistry, 21.06.2019 17:40
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 23.06.2019 02:00
Butane gas reacts with oxygen gas to give carbon dioxide gas and water vapor (gas). if you mix butane and oxygen in the correct stoichiometric ratio, and if the total pressure of the mixture is 390 mmhg, what is the pressure (in mmhg) of water vapor after the reaction is completed (temperature and volume do not change).
Answers: 2
You know the right answer?
How many grams of CO2 are dissolved in a 1.00 L bottle of carbonated water at 298 K if the pressure...
Questions

History, 21.10.2020 21:01


Spanish, 21.10.2020 21:01


Mathematics, 21.10.2020 21:01

Chemistry, 21.10.2020 21:01

Chemistry, 21.10.2020 21:01




English, 21.10.2020 21:01

Mathematics, 21.10.2020 21:01




Mathematics, 21.10.2020 21:01