
Chemistry, 17.03.2020 20:00 davfar334p47luq
Calcium carbonate is an "insoluble salt". But the solubility of CaCO3 can be increased substantially by acidifying the solution. Write all the relevant equilibrium equations and explain why adding acid will increase the solubility of calcium carbonate.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which of these sequences lists the correct order for the creation of sedimentary rock from sediment? a. deposition, burial, compaction, cementation b. burial, deposition, compaction, cementation c. compaction, deposition, burial, cementation d. cementation, deposition, burial, compaction
Answers: 1


Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1
You know the right answer?
Calcium carbonate is an "insoluble salt". But the solubility of CaCO3 can be increased substantially...
Questions


History, 24.03.2020 00:19











Business, 24.03.2020 00:20





Mathematics, 24.03.2020 00:20


History, 24.03.2020 00:21

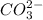 is converted to
is converted to 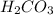 in acidic solution.
in acidic solution.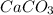 dissociates in solution to produce
dissociates in solution to produce 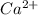 and
and 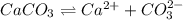
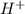 and gets converted to
and gets converted to 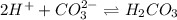 So,
So, 

