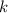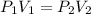A certain amount of chlorine gas was placed inside a cylinder with a movable piston at one end. The initial volume was 3.00 L and the initial pressure of chlorine was 1.85 atm . The piston was pushed down to change the volume to 1.00 L. Calculate the final pressure of the gas if the temperature and number of moles of chlorine remain constant

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1
You know the right answer?
A certain amount of chlorine gas was placed inside a cylinder with a movable piston at one end. The...
Questions






Computers and Technology, 22.04.2020 01:15

Mathematics, 22.04.2020 01:15


Mathematics, 22.04.2020 01:16

Mathematics, 22.04.2020 01:16


German, 22.04.2020 01:16


Mathematics, 22.04.2020 01:16



History, 22.04.2020 01:16

Arts, 22.04.2020 01:16

English, 22.04.2020 01:16

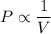
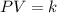 Pressure multiplied by volume equals some constant
Pressure multiplied by volume equals some constant