
Chemistry, 18.03.2020 18:55 Thelazysandwich
Based on the acid strength of H2SO3 and H2SeO3, will the equilibrium constant of H2SO3(aq)+HSeO3−(aq)⇌H2SeO3(aq)+HSO 3−(aq) be (Kc> 1) or (Kc< 1)? Group of answer choices Kc< 1, because H2SO3 is a stronger acid than H2SeO3 Kc< 1, because H2SO3 is a weaker acid than H2SeO3 Kc> 1, because H2SO3 is a weaker acid than H2SeO3 Kc> 1, because H2SO3 is a stronger acid than H2SeO3

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 06:00
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1


Chemistry, 23.06.2019 10:30
Which of the following pairs of elements is most likely to form an ionic compound? a oxygen and fluorine b sodium and aluminum c calcium and chlorine d nitrogen and sulfur
Answers: 1
You know the right answer?
Based on the acid strength of H2SO3 and H2SeO3, will the equilibrium constant of H2SO3(aq)+HSeO3−(aq...
Questions


Mathematics, 22.01.2021 02:40

Mathematics, 22.01.2021 02:40

Chemistry, 22.01.2021 02:40




Mathematics, 22.01.2021 02:40


Spanish, 22.01.2021 02:40

Chemistry, 22.01.2021 02:40



Mathematics, 22.01.2021 02:40

Mathematics, 22.01.2021 02:40



History, 22.01.2021 02:40


Biology, 22.01.2021 02:40

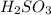 is a stronger acid than
is a stronger acid than 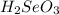

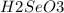 . Additionally, if we have his behavior the reaction would be displaced to the right side due to the strong acid (the strong acid is placed in the reactives side), so we will have a K greater than 1.
. Additionally, if we have his behavior the reaction would be displaced to the right side due to the strong acid (the strong acid is placed in the reactives side), so we will have a K greater than 1. 

