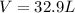Chemistry, 18.03.2020 20:05 Jadaaaaaaan
Determine the volume of SO2 (at STP) formed from the reaction of 96.7 g of FeS2 and 55.0 L of O2 (at 398 K and 1.20 atm). The molar mass of FeS2 is 119.99 g/mol. 4 FeS2(s) + 11 O2(g) → 2 Fe2O3(s) + 8 SO2(g)

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 06:00
Complete the sentences to best explain the ranking.match the words below to the appropriate blanks in the sentences.a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1

Chemistry, 23.06.2019 06:00
Nthis lab, you will do experiments to identify types of changes. using the question format you learned (shown above), write an investigative question that you can answer by doing these experiments
Answers: 3


Chemistry, 23.06.2019 10:30
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 3.75 mol fe and 8.70 mol nio(oh) react?
Answers: 1
You know the right answer?
Determine the volume of SO2 (at STP) formed from the reaction of 96.7 g of FeS2 and 55.0 L of O2 (at...
Questions

English, 20.08.2019 07:30

Chemistry, 20.08.2019 07:30

Biology, 20.08.2019 07:30

History, 20.08.2019 07:30





Mathematics, 20.08.2019 07:30

English, 20.08.2019 07:30


Mathematics, 20.08.2019 07:30

Mathematics, 20.08.2019 07:30


Mathematics, 20.08.2019 07:30

Mathematics, 20.08.2019 07:30



Mathematics, 20.08.2019 07:30

Biology, 20.08.2019 07:30

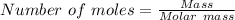
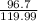
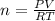

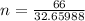
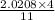 moles of FeS₂ will react with 2.0208 moles of oxygen to produce
moles of FeS₂ will react with 2.0208 moles of oxygen to produce 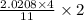 moles of SO₂
moles of SO₂
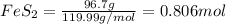
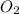
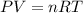
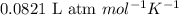
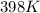
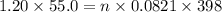
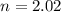
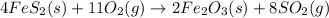
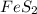
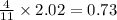 moles of
moles of 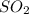
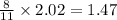 moles of
moles of 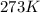 (at STP)
(at STP)