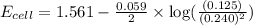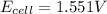Chemistry, 18.03.2020 21:41 cheerthi16
The standard reduction potentials for the Ag+|Ag(s) and Zn2+| Zn(s) half-cell reactions are +0.799 V and -0.762 V, respectively. Calculate the potential for the following electrochemical cell: Zn(s)|Zn2+(0.125 M)||Ag+(0.240 M)|Ag(s)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1

Chemistry, 22.06.2019 00:00
Several kinds of bears are found on earth. most bears are brown or black, but one type of bear, the polar bear, is white. what process led to this difference in fur color? explain your answer.
Answers: 1

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1
You know the right answer?
The standard reduction potentials for the Ag+|Ag(s) and Zn2+| Zn(s) half-cell reactions are +0.799 V...
Questions

History, 17.07.2019 12:00


Health, 17.07.2019 12:00

History, 17.07.2019 12:00

English, 17.07.2019 12:00


Health, 17.07.2019 12:00

Mathematics, 17.07.2019 12:00

Arts, 17.07.2019 12:00

Biology, 17.07.2019 12:00

Mathematics, 17.07.2019 12:00

Geography, 17.07.2019 12:00

Mathematics, 17.07.2019 12:00

English, 17.07.2019 12:00



History, 17.07.2019 12:00


Geography, 17.07.2019 12:00

Health, 17.07.2019 12:00

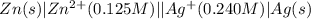
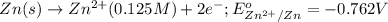
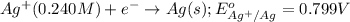 ( × 2)
( × 2)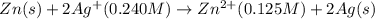
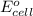 of the reaction, we use the equation:
of the reaction, we use the equation: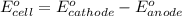
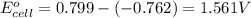
![E_{cell}=E^o_{cell}-\frac{0.059}{n}\log \frac{[Zn^{2+}]}{[Ag^{+}]^2}](/tpl/images/0552/5842/bffc2.png)
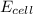 = electrode potential of the cell = ? V
= electrode potential of the cell = ? V![[Zn^{2+}]=0.125M](/tpl/images/0552/5842/c12f1.png)
![[Ag^{+}]=0.240M](/tpl/images/0552/5842/c82de.png)