
Chemistry, 18.03.2020 22:30 pumaben2864
In the laboratory you dissolve 22.8 g of chromium(III) acetate in a volumetric flask and add water to a total volume of 250 mL. What is the molarity of the solution? M. What is the concentration of the chromium(III) cation? M. What is the concentration of the acetate anion?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:10
The rock in a lead ore deposit contains 89 % pbs by mass. how many kilograms of the rock must be processed to obtain 1.5 kg of pb?
Answers: 1

Chemistry, 22.06.2019 00:00
How did planetesmals form planets? a. they broke apart into smaller chunks.b. they collided and stuck together.c. they cooled and pulled ice together.d. they began to rotate.
Answers: 1

Chemistry, 22.06.2019 19:30
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2

Chemistry, 22.06.2019 22:00
If a solution contains 3 moles/liter of sodium chloride (nacl, made of sodium ions and chloride ions), what is the osmolarity of this solution
Answers: 3
You know the right answer?
In the laboratory you dissolve 22.8 g of chromium(III) acetate in a volumetric flask and add water t...
Questions




Physics, 14.04.2021 21:10

English, 14.04.2021 21:10


Mathematics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10


Mathematics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10


Social Studies, 14.04.2021 21:10


Physics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10

Mathematics, 14.04.2021 21:10


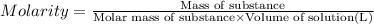
![[Cr(CH_3COO)_3]=\frac{22.8 g}{229 g/mol\times 0.250 L}=0.3982 M](/tpl/images/0552/7202/c3854.png)
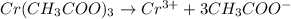
![[Cr^{3+}]=[Cr(CH_3COO)_3]=0.3982 M](/tpl/images/0552/7202/a4c9c.png)
![[CH_3COO^-]=3\times [Cr(CH_3COO)_3]=3\times 0.3892 M=1.1946 M](/tpl/images/0552/7202/97a2c.png)



