
Chemistry, 19.03.2020 00:48 yousifgorgees101
35.0 mL of stock hydrochloric acid solution is added to a 500 mL volumetric flask. A student adds distilled water up to the line. Using a pH meter and logarithms, he finds the molarity of the new solution to be 0.062 M HCl. What is the molarity of the stock solution?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 21:30
An atomic nucleus is composed ofa)protons.b)protons and neutrons.c)protons and electrons.d)protons, neutrons, and electrons.
Answers: 1

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2
You know the right answer?
35.0 mL of stock hydrochloric acid solution is added to a 500 mL volumetric flask. A student adds di...
Questions

Mathematics, 29.09.2019 02:30

Biology, 29.09.2019 02:30


History, 29.09.2019 02:30



Physics, 29.09.2019 02:30


Health, 29.09.2019 02:30

Physics, 29.09.2019 02:30



History, 29.09.2019 02:30


Mathematics, 29.09.2019 02:30

Social Studies, 29.09.2019 02:30

Mathematics, 29.09.2019 02:30

Mathematics, 29.09.2019 02:30

History, 29.09.2019 02:30

Advanced Placement (AP), 29.09.2019 02:30

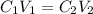
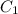 and
and 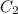 are initial and final concentration of a solution
are initial and final concentration of a solution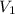 and
and 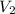 are initial and final volume of a solution
are initial and final volume of a solution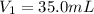 ,
, 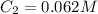 and
and 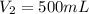
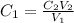 =
= 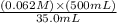 = 0.89 M
= 0.89 M

