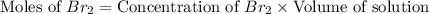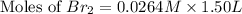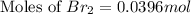Chemistry, 19.03.2020 01:16 blackwhiteroses383
Consider the reaction. 2 HBr(g) ¡ H2(g) + Br2(g) a. Express the rate of the reaction in terms of the change in concentration of each of the reactants and products. b. In the first 25.0 s of this reaction, the concentration of HBr drops from 0.600 M to 0.512 M. Calculate the average rate of the reaction during this time interval. c. If the volume of the reaction vessel in part b is 1.50 L, what amount of Br2 (in moles) forms during the first 15.0 s of the reaction?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1

Chemistry, 22.06.2019 10:30
Earth's axis of rotation is tilted at an angle of 23.5 degrees. what is one change you would see on earth if its axis was not tilted?
Answers: 3

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3
You know the right answer?
Consider the reaction. 2 HBr(g) ¡ H2(g) + Br2(g) a. Express the rate of the reaction in terms of the...
Questions


History, 07.10.2020 05:01

English, 07.10.2020 05:01


Mathematics, 07.10.2020 05:01

English, 07.10.2020 05:01

Social Studies, 07.10.2020 05:01


Biology, 07.10.2020 05:01

English, 07.10.2020 05:01


History, 07.10.2020 05:01

Mathematics, 07.10.2020 05:01


History, 07.10.2020 05:01


Mathematics, 07.10.2020 05:01


Health, 07.10.2020 05:01

History, 07.10.2020 05:01

![Rate=-\frac{1}{2}\frac{d[HBr]}{dt}=+\frac{d[H_2]}{dt}=+\frac{d[Br_2]}{dt}](/tpl/images/0553/1566/27c4e.png)
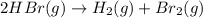
![\text{Rate of disappearance of }HBr=-\frac{1}{2}\frac{d[HBr]}{dt}](/tpl/images/0553/1566/d63dd.png)
![\text{Rate of disappearance of }H_2=+\frac{d[H_2]}{dt}](/tpl/images/0553/1566/eb73c.png)
![\text{Rate of formation of }Br_2=+\frac{d[Br_2]}{dt}](/tpl/images/0553/1566/30b3c.png)
![\text{Average rate}=-\frac{1}{2}\frac{d[HBr]}{dt}](/tpl/images/0553/1566/79555.png)
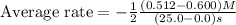
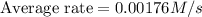
![\frac{d[Br_2]}{dt}=0.00176M/s](/tpl/images/0553/1566/78ef0.png)
![\frac{d[Br_2]}{15.0s}=0.00176M/s](/tpl/images/0553/1566/22daf.png)
![[Br_2]=0.00176M/s\times 15.0s](/tpl/images/0553/1566/5d9c4.png)
![[Br_2]=0.0264M](/tpl/images/0553/1566/42226.png)